Rockville, Maryland-based Hememics Biotechnologies Inc. has secured a $2.5 million investment from AMVI Partners, an international investment firm with offices in McLean, Virginia.
The startup is working on developing a handheld device equipped with a multiplex panel chip that is able to rapidly test a blood sample to detect bacteria or proteins in less than a minute. The initial device in development, named HEMEMICS, is a point-of-care panel to test for sexually transmitted diseases.
The company — one of the first of six health technology startups that the Northern Virginia-based Inova Health System invested in via its Inova Personalized Health Accelerator in 2017 — plans to use the investment to begin clinical research for a U.S. Food and Drug Administration submission for its flagship biotech product, it announced in a press release. Hememics connected with AMVI Partners through the Vietnamese American Business Council (VABC), a nonprofit based in McLean, Virginia, which has a mission to foster business opportunities between Vietnam and the U.S.
“Our long-term relationship with VABC has made this progress possible. This significant investment will enable us to achieve key milestones on our path to commercial availability,” said Hememics CSO and cofounder David Ho in a press release.
“We are thrilled to engage in this productive partnership with Vietnamese investors through AMVI Partners,” said Hememics CEO and cofounder John Warden in a statement. “Given the worldwide growth rates for STI such as chlamydia, gonorrhea and HPV, we are especially honored to have interest from the international investment community in our technology.”
The startup’s proprietary platform is protected by three patents, and the device doesn’t require refrigeration, according to its website. Here’s a visualization of how its technology works:
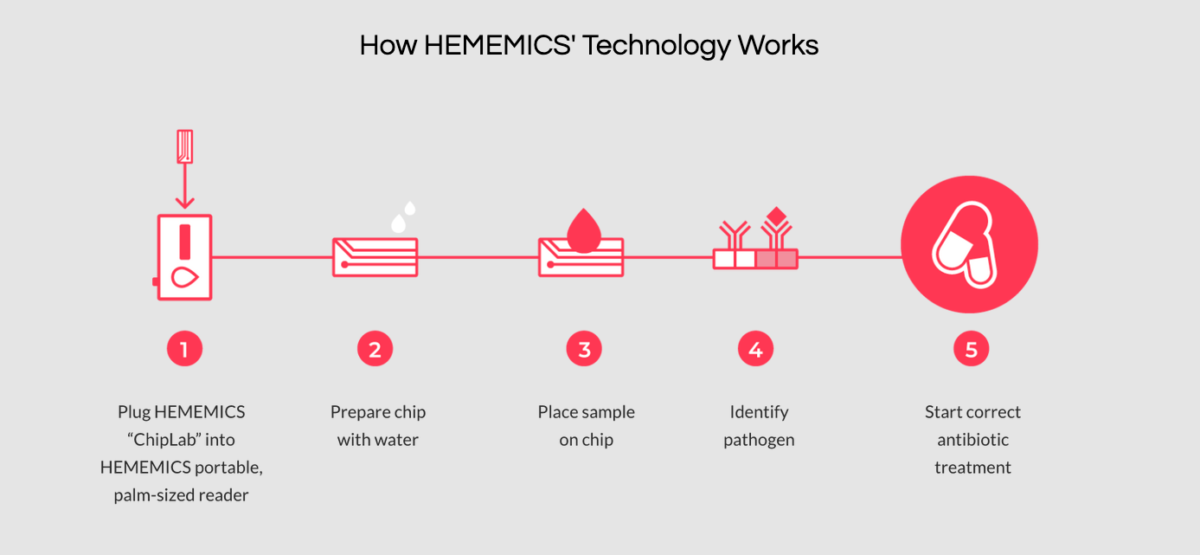
(Photo via Hememics Biotechnologis, Inc.’s website)
Before you go...
Please consider supporting Technical.ly to keep our independent journalism strong. Unlike most business-focused media outlets, we don’t have a paywall. Instead, we count on your personal and organizational support.
Join our growing Slack community
Join 5,000 tech professionals and entrepreneurs in our community Slack today!

The person charged in the UnitedHealthcare CEO shooting had a ton of tech connections

From rejection to innovation: How I built a tool to beat AI hiring algorithms at their own game

The looming TikTok ban doesn’t strike financial fear into the hearts of creators — it’s community they’re worried about


